JMLS 2021 June;6(1):31-37. Epub 2021 May 31
Copyright © 2021 by The Korean Society of Marine Life Science
Ingestion of Polystyrene Microplastics Acutely Induces Oxidative Stress in the Marine Medaka Oryzias javanicus
Sang-Eun Nam; Department of Marine Science, College of Natural Sciences, Incheon National University, Incheon 22012, Korea
Jee-Hyun Jung; Risk Assessment Research Center, Korea Institute of Ocean Science and Technology, Geoje 53201, Korea
Jae-Sung Rhee; Department of Marine Science, College of Natural Sciences, Incheon National University, Incheon 22012, Korea; Research Institute of Basic Sciences, Incheon National University, Incheon 22012, Korea; Institute of Green Environmental Research Center, Incheon 21999, Korea
- Abstract
Larvae from the marine medaka fish Oryzias javanicus were exposed with polystyrene microplastics (MPs) for 24 h. Exposure to waterborne fluorescent MPs showed clear ingestion and egestion in feces. Under constant MPs, the concentration of dissolved oxygen significantly decreased in 24 h compared to the control. Significant intracellular reactive oxygen species and malondialdehyde contents were detected in larvae, indicating oxidative stress and lipid peroxidation. Significant elevations in mRNA expressions of heat shock protein 70 and antioxidant defense system genes (glutathione reductase, glutathione peroxidase, catalase, and superoxide dismutase) were measured with increases in enzymatic activity of oxidative stress-related proteins. Taken together, the alterations to the molecular and biochemical components suggested that waterborne MPs had an oxidative stress effect on marine medaka larvae.
Keywords: Microplastics; Marine medaka; Oxidative stress; Antioxidant defense
Correspondence to: Jae-Sung Rhee; Department of Marine Science, College of Natural Sciences, Incheon National University, Incheon 22012, Korea; E-mail : jsrhee@inu.ac.kr
- Received
- 2 April 2021;
- Revised
- 14 April 2021;
- Accepted
- 8 May 2021.
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Microplastics (MPs) are widespread and ubiquitous emerging pollutants of aquatic ecosystems are reported globally in environ- mental components (e.g., air, soil, and water), due to a huge amount of annual production over 300 million tons (Andrady, 2011; Browne et al., 2011). Sixty percent of entire plastics produced to date was suggested to accumulate in the environment (Geyer et al., 2017). Plastic can be degraded to smaller pieces in aquatic environments by mechanical and/or photochemical forces, resulting in different characteristics compared to larger plastic such as small size, large surface area, and high surface curvature (Mattsson et al., 2015), which can result in the widespread con- tamination of aquatic environments. Manufactured small pieces of plastic such as cosmetics products (e.g. facial scrub, toothpaste, body wash) in a single use continue to be a growing area of concern (Napper et al., 2015). Freshwaters through emission from land to rivers are suggested as a main source for MPs pollution in marine environments (Besseling et al., 2017). Unique characteristics of MPs such as buoyancy and persistency lead their wide dis-persion through ocean currents from Polar regions to shorelines, beaches, seabed sediments, surface, and deep-sea (Thompson, 2015; Jamieson et al., 2019). Subsequently, the ubiquitous nature of MPs has become a major global environmental concern with highlighting potential biological interaction with aquatic biota and leads to public health problems (Ivar do Sul and Costa, 2014).
The majority of studies on MPs ingestion and their persistency to date have focused on marine animals (Setälä et al., 2014; Devriese et al., 2015). In numerous studies, MPs have been com- monly implicated in causing physiological damage to aquatic organisms at individual and/or population levels, such as gut blockage, oxidative stress, tissue damage, immunity modulation, reduced assimilation efficiency, leading to reduced fitness, neuro- transmission dysfunction, diminished energy metabolism, retarded growth or mortality (Barbosa et al., 2020; Gouin, 2020). Further- more, the potential toxic effects of MPs have consistently been suggested in fish. However, there are also reports on no acute effects of MPs to mobility or survival of aquatic animals and even high concentrations of MPs tested showed no observable detrimental effects (Hämer et al., 2014; Kaposi et al., 2014; Weber et al., 2018). Thus, studies on the biological impact of MPs should consistently be conducted on numerous animal taxa including fish.
In this study, we aimed to measure acute molecular and bio- chemical responses of the marine medaka (Oryzias javanicus) larvae exposed to waterborne polystyrene MPs. The marine medaka O. javanicus is widely distributed in the coastal regions of East to Southeast Asia (Inoue and Takei, 2002) as a promising test organism for ecotoxicological studies due to relatively small size (3~4 cm), a short generation period (3~4 months), daily spawning and transparent embryos, sexual dimorphism, and ease of mass culture in the laboratory (Kim et al., 2018; Nam et al., 2020). The molecular and biochemical responses of fish are extensively employed for estimating the potential effects of exogenous con- taminants. Thus, to understand the potential acute effects of MPs, several molecular and biochemical responses were analyzed in the present study. Polystyrene microbeads were chosen as numerous studies have suggested their ingestion in aquatic organisms and thus, comparative study is able in the field. The relatively higher density of polystyrene than water hence makes it to rise and sink in aquatic environment (Driedger et al., 2015). In particular, several important ecotoxicological endpoints, the cell stress (hsp70) and oxidative stress parameters such as intracellular reactive oxygen species (ROS), malondialdehyde (MDA), and mRNA expression and enzymatic activity of antioxidant defense system [glutathione reductase (GR), glutathione peroxidase (GPx), catalase (CAT), and superoxide dismutase (SOD)] were analyzed in response to water- borne MPs for 24 h.
Materials and Methods
1. Marine medaka
The Java medaka O. javanicus was cultured under controlled conditions at 26℃ with a light:dark (L:D) photoperiod of 14 L:10 D in artificial seawater (ASW; TetraMarine Salt Pro, TetraTM, Cincinnati, OH, USA; 6.48±0.59 mg O2 L-1; pH 7.8~8.1) adjusted to 12 prac- tical salinity units (PSU). The fish were reared in glass aquaria (60 ℓ capacity; 21c-HighTech®, Pusan, South Korea) with aeration and each glass aquaria accommodated up to 28 adult fish (both sexes). They were fed Artemia salina (< 6 h after hatching; SERA Artemia, Salt Lake, UT, USA) twice a day until satiation.
2. Microplastics exposure
To confirm the ingestion of MPs by the fish larvae, 1 μm of fluorescent microparticles based on melamine resin (carboxylate-modified; fluorescein isothiocyanate (FITC)-marked; analytical stand- ard) was purchased from Sigma-Aldrich, Inc. (St. Louis, MO, USA). For exposure, non-functionalized polystyrene microbeads (1 μm) were purchased as an aqueous suspension (2.5 mg ml-1) from Sigma-Aldrich Inc. According to the manufacturer, the mean diameter of the particle was 1 μm, and the calibrated particle diameter was 1.00~1.05 μm. The suspensions were gently mixed using a vortex before dilution and use. No surfactant was used during the dilution procedure. Working solutions were prepared for designated nominal concentrations by dilution of the stock solution in artificial seawater (12 PSU). For control and exposure, 90 larvae were prepared and separated into three groups (30 fish per group) as triplicate experiments. The 30 fish were reared in 1.5 ℓ chamber (length 280 × width 65 × depth 160 mm) of aquaria (21c-HighTech®) with 100 particles ml-1 for 24 h. Dissolved oxygen was monitored at 0, 1, 3, 6, 9, 12, 15, 18, 21, and 24 h using a Orion Star meter (520M-01A, Thermo Fisher Scientific Inc., MA, USA). At the same time, total ammonia nitrogen (TAN), nitrite- nitrogen (NO2-N), and nitrate-nitrogen (NO3-N) were analyzed at by stopping inflow of the ASW for 24 h. Standard analytical methods were used to measure TAN, NO2-N, and NO3-N (Grasshoff et al., 1983).
3. Measurement of intracellular reactive oxygen species and lipid peroxidation
After exposure to waterborne MPs, pooled larvae were hom- ogenized in a buffer containing 0.32 M sucrose, 20 mM HEPES, 1 mM MgCl2, and 0.5 mM PMSF (pH 7.4) using a Teflon hom- ogenizer. The homogenate was centrifuged at 10,000 × g for 20 min at 4℃ and the supernatant was collected and analyzed. Black plates with 96 wells were filled with phosphate-buffered saline buffer, the probe (H2DCFDA at a final concentration of 40 μM), and the supernatant, which had been fractioned to a final volume of 200 μl. Measurements were obtained at an excitation wavelength of 485 nm and an emission wavelength of 520 nm using a ThermoTM Varioskan Flash (Thermo Fisher Scientific, Tewksbury, MA, USA) and they were normalized using the total protein values. The protein levels were measured using the Bradford method. MDA contents were measured using the fol- lowing method: the samples were pooled and homogenized with a Teflon homogenizer in five volumes of Tris buffer (20 mM) con- taining NaCl (150 mM), aprotinin (2 μM), benzamidine (100 μM), β-mercaptoethanol (10 mM), and leupeptin (20 μM). The samples were centrifuged at 30,000 × g at 4℃ for 30 min and the resulting supernatants were heat-denatured at 75℃ for 15 min. Thiobarbituric acid reactive substances were quantified at 535 nm using a Thermo Varioskan Flash spectrophotometer (Thermo Fisher Scientific). The thiobarbituric acid reactive substance con- centrations were measured using a standard curve based on MDA bis (dimethylacetal, Sigma-Aldrich, Inc.). Then, the MDA levels were determined using a calibration curve and calculated as nM of MDA per μg of the total sample.
4. Total RNA extraction
Total RNA was extracted using TRI Reagent (Molecular Research Center Inc., Cincinnati, OH, USA) according to the manufacturer's protocol and DNA digestion was performed using DNase I (Sigma-Aldrich). Total RNA was quantified by light absorption at A260 and quality checked by analyzing the A230/260 and A260/280 ratios using a NanoDrop ND-1000 spectrophotometer (Thermo Scientific, Wilmington, DE, USA).
5. Real-time polymerase chain reaction
Transcriptional changes were measured using a real-time poly- merase chain reaction (PCR) and the previously confirmed primer sets for marine medaka (Kim et al., 2018; Nam et al., 2020). Exactly 2 μg of total RNA was used to synthesize the cDNA. Real-time (RT)-PCR was conducted using SYBR® Green Master Mix (Bio-Rad, Hercules, CA, USA). The reactions were run at a final volume of 25 μl and the sample contained the following components: 12.5 μl of SYBR Green buffer, 1 μl each of forward and reverse primers (250 nM), 9.5 μl nuclease-free water, and 1 μl cDNA (total RNA equivalent). All RT-PCR experiments were performed in unskirted low 96-well clear plates (Bio-Rad). Amplification and detection of SYBR Green-labeled amplicons were performed under optimized conditions using the CFX96 real-time PCR system (Bio-Rad) under the following conditions: 95℃ for 5 min; 40 cycles of 95℃ for 20 s, 55℃ for 30 s, and 72℃ for 40 s. The cycles were continued to determine the melting curve to confirm the amplifi- cation of specific products. Data from the triplicate experiments were expressed relative to the expression of the internal control β-actin gene that was used to normalize for any differences in the reverse transcriptase efficiency. Each transcriptional level was determined using the 2-ΔΔCt method.
6. Measurement of antioxidant defense system enzyme activities
The activities of enzymes involved in the antioxidant defense system (i.e., glutathione S-transferase, GST; glutathione peroxidase, GR; catalase, CAT; and superoxide dismutase, SOD) were measured according to our previous studies on marine medaka (Kim et al., 2018; Nam et al., 2020). The sample was rinsed and homogenized in trichloroacetic acid and cold buffer (0.25 M sucrose, 10 mM Tris, 1 mM EDTA, 0.2 mM DTT, and 0.1 mM PMSF; pH 7.4) at a ratio of 1 to 10 (w/v) using a Teflon homogenizer. The hom- ogenates were centrifuged at 3,000 to 10,000 × g for 10 min at 4℃ and then the upper aqueous layer was collected for enzymatic assay according to the manufacturer's protocol. The GR activity was measured using a glutathione reductase assay kit (Catalog No. GRSA; Sigma-Aldrich, Inc.) and the CAT and SOD activities were measured using catalase (Catalog No. CAT100; Sigma-Aldrich Chemie, Switzerland) and SOD assay kits (Catalog No. 19160; Sigma-Aldrich Chemie) at 25℃ and absorbances of 520 and 440 nm, respectively. The oxidative and antioxidant parameters were measured using a spectrophotometer (ThermoTM Varioskan Flash), normalized using the total protein values, and shown as a percentage of the control. Total proteins were determined using the Bradford method.
7. Statistical analysis
Statistical analysis was undertaken using the SPSS statistical software (ver. 17.0; SPSS Inc., Chicago, IL, USA) and the data were expressed as the mean ± standard deviation (S.D). Significant differences between the control and treatment groups were analyzed using one-way analysis of variance followed by Tukey's test at the p < 0.05 significance level.
Results and Discussion
Since marine ecosystems are highly affected by plastic debris daily, the effects of plastic pollutants should consistently be studied on the molecular/biochemical response and physiology of marine fish. In this study, MPs were ingested by marine medaka larvae, and its fluorescence was clearly detected in the digestive system of the fish (Fig. 1). Retention times of MPs in digestive varied in the tested marine medaka. Several studies have shown that medakaspecies ingest different types and sizes of MPs (Cong et al., 2019; Wang et al., 2019; Assas et al., 2020). Although there are conflicting studies on whether MPs can be permanently accumulated in fish organs instead of egestion, temporarily accumulated MPs and their movement in the digestive system were observed in this study. Compared to the control, respiratory rate of MPs-exposed larvae tended to be increased for 24 h with increasing exposure period (p < 0.05) (Fig. 2A), suggesting MPs-triggered respiration stress in the fish. The concertation of NH4-N as one of the markers for ammonia excretion showed no significant modulation during MPs exposure (Fig. 2B). Since the respiration rate in fish is directly associated with gill function (e.g., ventilation, perfusion, gas exchange efficiency), increasing respiration would suggest imbalance in arterial O2 content, energy expenditure due to O2 availability, and inevitable consequence of acute stress by MPs exposure. Thus, further experiment was conducted to investigate MPs-triggered oxidative stress and responses of antioxidant defense system.
The MPs treatment significantly increased intracellular ROS and MDA levels for 24 h (p < 0.05) (Fig. 3A and 3B). The greater lipid peroxidation observed after 12 h-exposure to the MPs was con- sistent with the increased intracellular ROS level. The measurement of modulation in intracellular ROS level is a strong biomarker that can be used to expect acute oxidative stress and the subsequent cellular damage. MDA is one of the final products through lipid peroxidation and/or damage on mitochondria due to the suscep- tibility of lipids to oxidation, which are strongly related to ROS formation (e.g., hydroxyl radicals, superoxide radicals, and hydrogen peroxide) in cells.
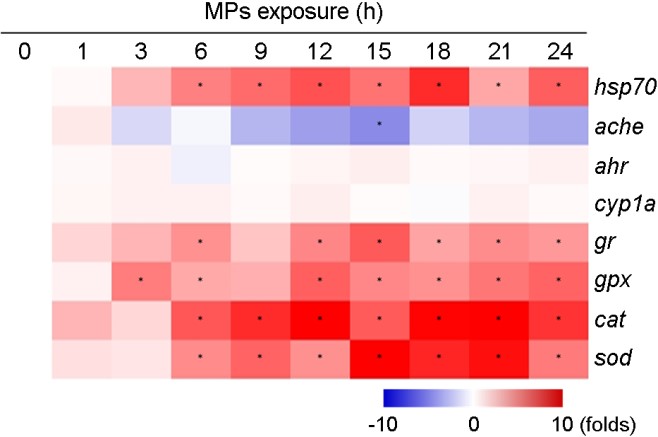
The mRNA of stress protein hsp70 was highly sensitive to MPs in the marine medaka larvae, as the fish exposed to MPs showed significant induction of hsp70 mRNA compared to the control (p < 0.05) (Fig. 4). Basically, Hsp70 protein as a molecular chaperone is expressed in response to a variety of exogenous/endogenous stressors and maintain homeostasis by protecting crucial cellular functions in most organisms (Feder and Hofmann, 1999). There- fore, increase in mRNA expressions of hsp70 upon MPs exposure suggests that MPs would be an acute stressor on the marine medaka larvae and the fish employs hsp70 to maintain homeostasis by protecting cells from MPs-triggered damage.
In this study, there was a significant increase in the mRNA expressions of essential components including GR, GPx, CAT, and SOD in antioxidant defense system in marine medaka larvae in response to MPs exposure for 24 h (p < 0.05) (Fig. 4). Measure- ment of modulation in antioxidant defense system has suggested the total oxyradical scavenging capacity in fish (Regoli et al., 2002). Of the members, GR protein catalyzes the reduction of GSSG to GSH form for the provision of GSH into the redox cycle and GPx enzyme plays an essential role in detoxifying hydrogen and lipid peroxides by incorporating GSH (Lesser, 2006). The enzymes CAT and SOD play a crucial role in cellular defense mechanisms upon oxidative stress by catalyzing the dismutation of superoxide anions to hydrogen peroxide and molecular oxygen (Lesser, 2006). The SOD intrudes into the first transformation by the dismutation of the superoxide free radicals O2- into H2O2, and CAT converts it into H2O and O2. Thus, the induction of the antioxidant defense system in the MPs-exposed marine medaka larvae suggests that the fish would maintain homeostasis against MPs-triggered acute oxidative stress through the inhibition of oxidative stress by stimulating the defense system, as sensitivity and capacity of the antioxidant defense system are generally associated with the health status in animals. There was no significant modulation in mRNA expressions of the detoxification metabolism-related genes ahr and cyp1a1 (p > 0.05) (Fig. 4). Since the phase I/II biotransformation is activated through direct binding of xenobiotics to the ligand-activated transcription factor AhR (Hahn et al., 1998), we assume that MPs are not responsible for AhR-mediated detoxification metabolism for 24 h exposure. Further studies on long-term treat- ment are required to clarify because metabolites or extracts of MPs can be associated with the induction of biotransformation.
The enzymatic activities of the first line of defense against oxidative stress, CAT and SOD, and the GSH regulators, GR and GPx were significantly elevated by exposure to MPs (p < 0.05) (Fig.5). Enzymatic activities of antioxidant defense system strongly corroborated the findings from mRNA expressions of the system because mRNA expression is strongly associated with protein synthesis and activity. Taken together, it can be concluded that the drastic increase in the antioxidant defense system response is due to the acute oxidative stress induced by MPs exposure through excessive intracellular ROS and MDA production in the marine medaka larvae. The results obtained in this study also suggest that the increased prevalence of MPs in marine environments will have potential effects on early developmental stages of aquatic vertebrates via modulation in its ability to strongly regulate antioxidant systems.
- References